 
Negative beta decay is far more common than positive beta decay. The neutrino and the antineutrino are high-energy elementary particles with little or no mass and are released in order to conserve energy during the decay process. One type (positive beta decay) releases a positively charged beta particle, called a positron, and a neutrino the other type (negative beta decay) releases a negatively charged beta particle, called an electron, and an antineutrino. Beta decay occurs when an atom has either too many protons or too many neutrons in its nucleus. This is why plutonium, with a long half-life, is extremely hazardous if ingested.Ītoms emit beta particles through a process known as beta decay. A 4 MeV alpha particle can only travel a few centimetres through the air.Īlthough the range of an alpha particle is short, if an alpha decaying element is ingested, the alpha particle can do considerable damage to the surrounding tissue. Since an alpha particle is relatively more massive than other forms of radioactive decay, it can be stopped by a sheet of paper and cannot penetrate human skin. Mass number decreases by 4, atomic number decreases by 2 (due to loss of 2 protons and 2 neutrons).Īlpha decay usually occurs in heavy nuclei such as uranium or plutonium, and therefore is a major part of the radioactive fallout from a nuclear explosion. Uranium 238 decays by alpha emission to give Thorium 234 The original radionuclide is called the parent and the new radionuclide produced after decay is called the daughter product. 
the totals are the same before and after the decay. In the following equations, both mass number and atomic number are conserved, i.e. Representation of decay by symbols and equations Unstable nuclei are called radioisotopes or radionuclides. 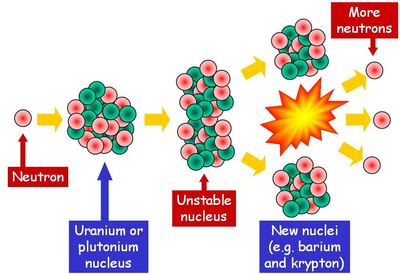
The release of energy and/or matter allows unstable nuclei to achieve stability. Radioactive decay is the breakdown of a nucleus to release energy and matter from the nucleus. An isotope is identified by specifying its chemical symbol along with its atomic and mass numbers. the same atomic number but a different mass number. An isotope of an atom has the same number of protons but a different number of neutrons, i.e. It is possible to have different versions of the same element, called isotopes. In a chemical symbol for an element it is written as a superscript before the element symbol.Įxample: One type of atom of uranium has 235 nucleons so we write 235UĮach element in the periodic table has a different atomic number and is identified by that number. The mass number, A, is the number of nucleons in a nucleus. In a chemical symbol for an element it is written as a subscript before the element symbolĮxample: There are 92 protons in the nucleus of a uranium atom so we write 92U The atomic number, Z, equals the number of protons in the nucleus. To examine nuclear reactions it is necessary to define a number of terms used to describe a nucleus.Ī nucleon is a particle in a nucleus, i.e. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
